
Eli Lilly and Boehringer Ingelheim Cancel Investments
A clear signal to Berlin: the pharmaceutical companies Eli Lilly and Boehringer Ingelheim are canceling planned investments in Germany.

A clear signal to Berlin: the pharmaceutical companies Eli Lilly and Boehringer Ingelheim are canceling planned investments in Germany.

Sygnature Discovery has partnered with AI firm DaltonTx to sharpen its drug discovery pipeline, bringing faster medicinal chemistry decisions and tighter data privacy to its CRO offering.

Northway Biotech is finalizing the Baltic region's most advanced cell therapy center — a 3,500 m² facility at Vilnius City Innovation Industrial Park, set to open Q3 2025.

Precise, reproducible, and regulatory-ready: CE-based methods are redefining how scientists characterize therapeutic mAbs and ADCs across the biotherapeutic development pipeline.
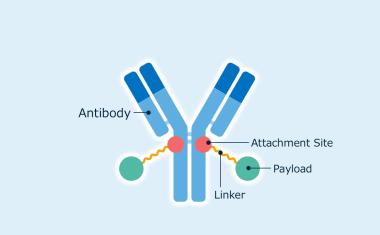
Asahi Kasei Life Science has acquired the license rights for a novel antibody-drug conjugate (ADC) technology from The Noguchi Institute.

Acquisition strengthens US peptide manufacturing capabilities and broadens global expertise in complex peptide APIs

Catalent partners with non-profit Elpida Therapeutics to support late-phase AAV manufacturing for a rare neurological gene therapy, securing exclusive rights across Elpida's broader AAV pipeline.

Hongene Biotech delivers end-to-end CDMO services for SiranBio's dual-target siRNA hepatitis B candidate, from raw materials through cGMP manufacturing, highlighting its capabilities in oligonucleotide therapeutics.

Bristol Myers Squibb licenses Lonza's SYNtecan linker-payload platform to advance an undisclosed ADC target, with Lonza eligible for upfront payments, milestones and royalties on resulting products.

Piramal Pharma Solutions opens a new payload-linker suite in Michigan as part of a $90 million US investment plan, bolstering its ADC manufacturing capabilities across sites in Riverview and Lexington.

Symeres, a transatlantic CRDMO for drug discovery and development, announced the appointment of Henning Steinhagen as Chief Executive Officer.

Takeda has officially commissioned a new photovoltaic system at its production site in Oranienburg near Berlin.

Explore how pellet-based systems can de-risk development and scale-up of robust modified-release formulations across the product lifecycle.

All closing conditions satisfied for the acquisition of three small molecule sites in the US and Australia; closing to occur as of May 1, 2026. Adds highly demanded US-based manufacturing capacity to Siegfried's network.

In this CHEManager Leaders & Motivators interview, Frank Romanski, Lonza Capsugel, reflects on how strategy becomes execution and what resilience demands from teams,

India’s CDMO sector is rapidly advancing: stronger R&D, complex chemistry, digitalization, and efficiency gains. Astec boosts this with new R&D centers, automation, and global‑standard operations.

The investment, its largest since its inception, demonstrates continued progress against AbbVie's $100 billion commitment to US R&D and capital investments, including manufacturing, over the next decade.

Evonik is expanding its biotechnological production capacities at the Fermas site in Slovenská Ľupča with an investment of around €80 million

Asahi Kasei has completed its acquisition of Germany’s Aicuris, adding three antiviral assets, in a deal previously reported at about €780 million.

Acquisition includes industry-leading ADC platform, its clinically-validated tubutecan linker-payload technology, and next-generation platform to expand Gilead’s ADC capabilities.

Merck has launched its acquisition of Terns Pharmaceuticals, a deal valued at about $6.7 billion that would add to its hematology portfolio.

Ofichem has unified its six brands under the single Ofichem name, creating a fully integrated, family-owned CDMO and pharmaceutical supplier.

On Demand | Join biopharma professionals, CDMO leaders, and supply chain innovators for a virtual event exploring the future of outsourced biomanufacturing.

Adragos Pharma has acquired Sanofi’s Maisons-Alfort sterile fill-finish site in France, expanding its European manufacturing network and prefilled syringe capabilities.

The acquisition marks Samsung Biologics’ geographical diversification, establishing its first US manufacturing presence and expanding regional client support.

When international companies bring a new drug or medical device to the European market, they face many challenges.

As discovery accelerates and throughput rises, digital transformation is becoming essential for chemistry labs. AI, robotics, and data growth must be orchestrated to boost productivity—rather than overwhelm the lab.
AI and evolving regulations set to transform biopharma operations in drug development and quality.

At CPhI Frankfurt, Christene Smith of CHEManager International interviews Axplora CCO Arul Ramadurai on navigating industry uncertainty and building strategic partnerships.

From In Silico Design to In Vitro Impact

LGM Pharma invests an additional $9 million to expand US manufacturing, bringing total investment to $15 million across its Texas and Colorado facilities.

Ice Pharma recently announced a strategic expansion beyond its core API business, positioning the company to compete in higher-value finished dosage, nutraceutical, and specialty excipient markets.

First Clariant facility in the United States to manufacture pharmaceutical-grade polyethylene glycol excipients for oral and topical applications.

Full-building, 15-year lease for 64,000 square feet enables CDMO CordenPharma Colorado to power next-generation peptide development in Boulder, Colorado.

$60 million investment to expand highly potent API (HPAPI) development and manufacturing at Axplora’s Farmabios site in Gropello Cairoli, Italy.



























