Axplora Expands Capabilities at Le Mans Site
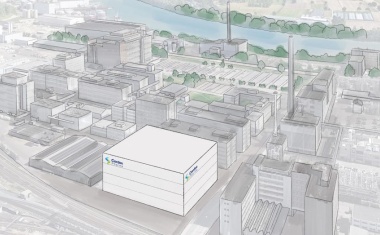
Axplora, an API small molecule and ADC (antibody-drug conjugate) manufacturer, is strengthening its position in the commercial manufacturing ADCs by launching a cutting-edge payload manufacturing workshop at its Le Mans site (France).