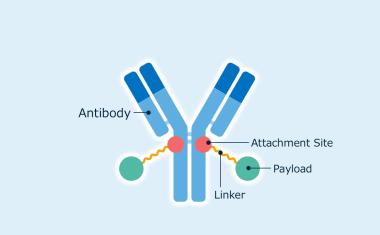
Northway Biotech Invests €61 Million in Vilnius Cell Therapy Manufacturing Center
Northway Biotech is finalizing the Baltic region's most advanced cell therapy center — a 3,500 m² facility at Vilnius City Innovation Industrial Park, set to open Q3 2025.