Insys Files for Chapter 11 After Fines
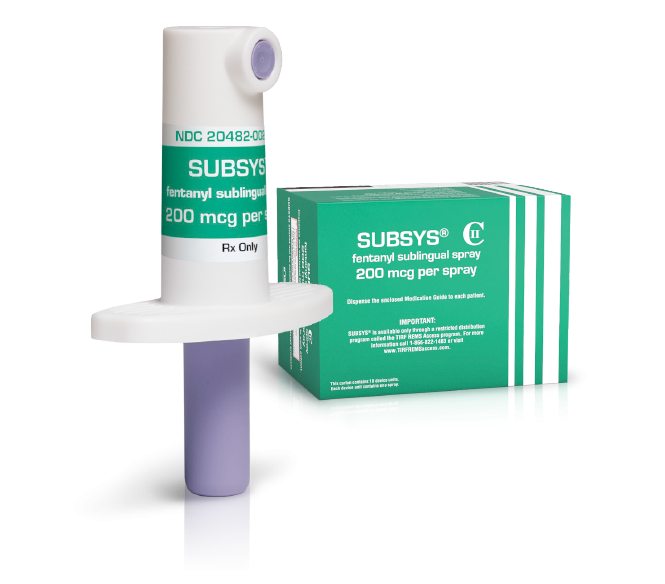
The growing US opioid crisis has claimed its first corporate victim. Drugmaker Insys Therapeutics filed for Chapter 11 bankruptcy protection last week, a few days after pleading guilty to federal fraud charges and agreeing to pay $225 million to settle allegations tied to its fentanyl-based painkiller Subsys.
Insys, which is based in Arizona, said it would use the bankruptcy process to arrange an asset sale within 90 days without interrupting payments to vendors and suppliers or interfering with normal operations.
“After conducting a thorough review of available strategic alternatives, we determined that a court-supervised sale process is the best course of action to maximize the value of our assets and address our legacy legal challenges in a fair and transparent manner,” said CEO Andrew Long.
The drugmaker has pleaded guilty to five counts of mail fraud after authorities alleged it had paid kickbacks and pursued unlawful marketing of Subsys. Four former executives were accused of scheming to bribe doctors into prescribing the drug used to treat pain in certain adult cancer treatments.
The case is thought to be the first conviction of pharmaceutical company CEO on charges related to opioids. Lawsuits have claimed that in an executive-driven bribery scheme doctors were treated to expensive seafood dinners and strip club visits as part of a sham speaker program directed by the company’s marketing team.
Along with top executives, reports say several former Insys sales managers have been implicated in the scheme, along with five New York City doctors.











