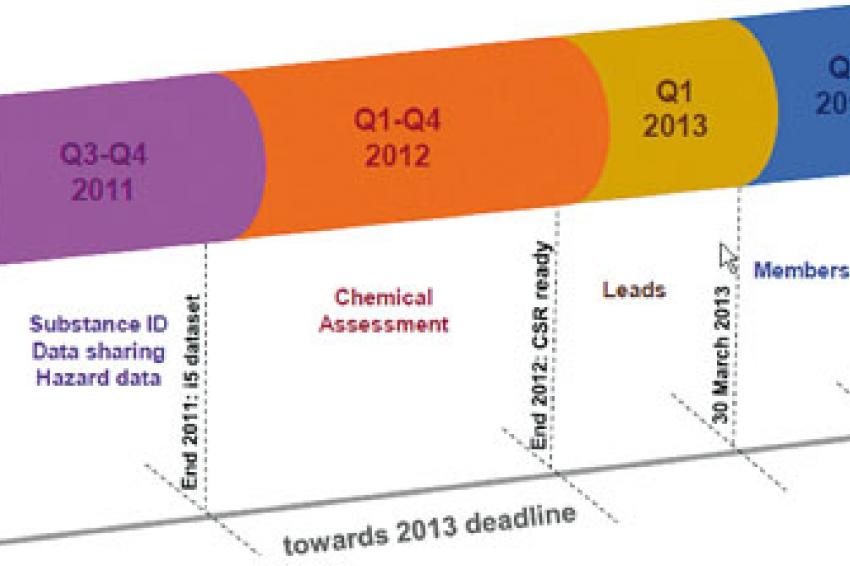
Reach In 2013
What Challenges Can Be Avoided in the Next Registration Wave
First Round - By Dec. 1, 2010, the European Chemical Agency (ECHA) had received approximately 25,000 registration dossiers for about 4,700 different chemical substances that are produced in or imported into the European Union, mainly in the tonnage band of over 1,000t/y.
Furthermore, it received more than 3.1 million notifications from approximately 110,000 different substances where basic classification data were communicated. It has been estimated that this huge effort has cost the European chemical industry already more than €1.2 billion.
Was this a success? How have the companies managed to get there?
Preparing For Reach:
Four months before the first registration deadline, the EU's Directors' Contact Group on Reach Feedback Advocacy summarized the main difficulties of the process like this:
- SIEF problems
- Completeness of dossiers
- Dependency on the Lead Registrant
- Uses not covered by a registration
- Legal entity change
- Confidentiality of substance name for classification and labeling inventory
- Stability of guidance
For the next registration wave (for substances in the 100-1,000t/y tonnage band), it is very likely that these challenges will occur again if we do not succeed in transferring the experiences of the first wave in an appropriate way. The crucial step for this success is an efficient and streamlined cooperation in the Substance Information Exchange Forum (SIEFs). But to come to this level, each company has to prepare itself according to a number of basic rules to be ready to contribute to the SIEFs:
5 Golden Rules For Basic Reach Preparation:
- 1. Close cooperation through industry associations is strongly recommended as they are aware of all basic aspects of Reach and Classification, Labeling and Packaging (CLP).
- 2. Budgeting is the financial prerequisite for a change. Estimation of costs as a result of Reach and CLP is difficult and continuously changing, but there is good guidance available via Cefic and ECHA as well as through associations and consultancies. Budget adequately and on time.
- 3. Use the full time frame. Concentrate on substances with the highest priority; develop an in-house Reach/CLP strategy; allocate resources accordingly.
- 4. Communication is crucial. Internally, create awareness for all affected departments such as marketing, sales, procurement. Externally, create transparency for your customers and suppliers. For example, create a company-specific homepage for Reach.
- 5. Start on time (and check your schedule regularly.). Adopt your IT/ERP systems and implement the changes in the production process.
Efficient Cooperation In SIEFs:
Which tasks are expected from a SIEF - and which are out of scope?
In Reach legislation there are very clear descriptions of the expected achievements of a SIEF:
- Substance Sameness ("OSOR," one substance, one registration.)
- Classification and labeling
- Data availability - data gap analysis
- Lead registrant (LR) vote
- Regular progress status report
- Joint/separate chemical safety report (CSR)
- Joint dossier submission
The members of a SIEF can freely decide whether or not they will create a consortium, where the more active members are grouped and transfer these obligations accordingly. A consortium is by no means a legal requirement. In practice, it has turned out that the consortium is the most powerful force in the registration process. Where ever applicable, it is strongly recommended that a consortium be created. Here again are some hints to be considered:
- SIEF communication needs to be consistent and efficient.
- Use existing reference documents and contract templates (e.g. CEFIC, other trade associations). Don't re-invent the wheel.
- SIEF management can be done by LR or outsourced to service providers.
- An appropriate SIEF communication platform is the key. Depending on the size of the SIEF, it is sufficient to communicate on just an email-level (avoid cumbersome and expensive IT tools.)
Main Issue: The Costs
The regulation states: "Cost should be transparent, fair and non-discriminatory." This is not very helpful. Nonetheless, the main cost drivers of a working SIEF/Consortium are well known:
- SIEF management
- Preparation of IUCLID dossiers, exposure scenarios, chemical safety reports, risk management measures, etc.
- Generation of invoices, letters of access (LoA), etc.
- Provision for reimbursement (limits should be agreed in the contracts.)
The more transparent the costs and the sooner they are communicated within a SIEF, the less misunderstandings will occur. For active SIEF members and for the LR in particular, some basic rules regarding costs apply:
- Pay only for the data you need (tonnage band, intermediate ...).
- Explain the cost sharing system in an easily understandable way.
- Give an early cost-estimate if possible.
- Communicate clearly at which point in time SIEF members will get the data, the LoA, the invoice, etc.
Details should be regulated in the SIEF agreement as mandatory basis for cooperation. Cefic published notes on transparency and fair cost sharing.
What Can Happen During the Cooperation in SIEFs?
Even though the publically available standard contract templates (consortium agreement, SIEF agreement, LoA, etc.) that were prepared by the legal experts of Cefic to cover most scenarios, lots of unpredictable things can happen when SIEFs work together:
- Member opts out and blames the consortium for unfair or discriminatory costs
- Member becomes bankrupt after having signed the SIEF agreement
- Member doesn't communicate CSR-requiring uses due to company owned IP
- Member doesn't handle data confidentially
- Member acts in a non-compliant manner with regard to competition laws
Experience shows that also the LR is not on the safe side:
- LR becomes bankrupt after having agreed his role
- LR does not submit a registration dossier in time
- LR submits his dossier, but subsequently fails technical completeness check
- Legal entity/owner change (also for members)
Not every possible scenario can be regulated. In these cases SIEFs have to decide individually what to do - ideally in a unanimous way.
What Will Be Different in 2013?
On the one hand, the European industry has had a lot of experiences during the first registration wave. Provided these can be communicated effectively to new registrants, the second and third registration waves should be accomplished much more smoothly.
On the other hand, there are clear indications that the upcoming registrations will be more difficult:
- Average number of SIEF members will decrease (fewer companies per substance)
- Expect fewer substances to be handled in consortia
- More small and medium-sized enterprises will require registrations
- SIEF management challenges will become even more important
- Substances are likely to be data poor
- Less experienced LRs will be managing SIEFs
Conclusion
Most of the European chemical manufacturers/importers will be obliged to register their substances in future - many of them for the first time.
The first registration wave in 2010 demonstrated that this pro¬cess is manageable when a few basic hints are followed:
- Start your registration process now.
- Check if the substance was already registered in 2010 and contact LR/consortium.
- If not registered yet, consider forming consortium/SIEF leadership team.
- Do the necessary basic SIEF steps ASA.
- Use documents and tools available via CEFIC website.
- ... and finally: make use of existing experience.
Good luck for 2013!
Contact
Linde AG
Seitnerstr. 70
82049 Pullach
Germany
+49 89/7446-0
+49 89/7446-1144