Winning Strategies in the Pharmaceutical CDMO Industry
How Companies Cope with Geopolitical Risks and Mechanistic Changes in Pharma Supply
Despite the favorable economic environment, the global economic risk factors have intensified against the background of record global debt and a rise in geopolitical risk. Amid growing uncertainty and regionalization, values such as trust, reliability, consistency, and cultural and regional proximity continue to gain importance.
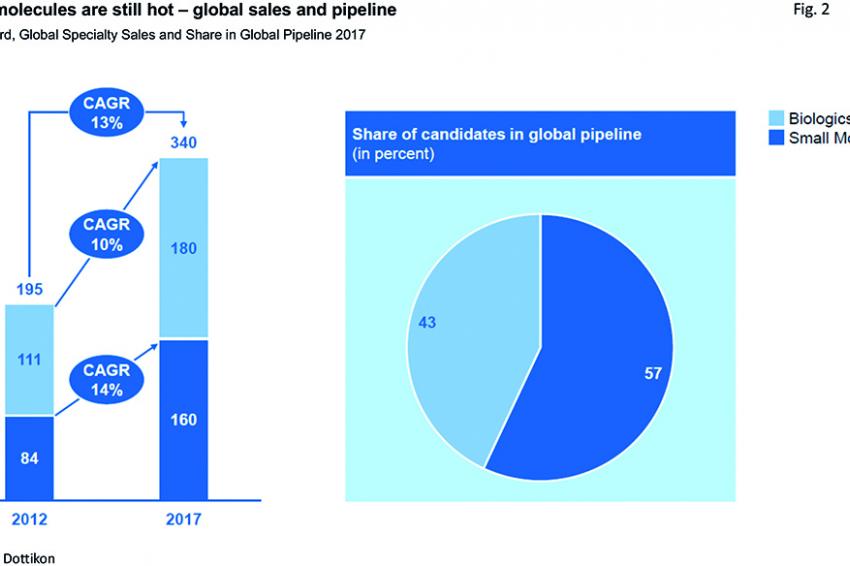
The demographic developments continue to ensure further long-term volume growth in the pharma market. An increase in the number of older people with higher life expectancy in industrialized countries, environmental pollution, and an increase in wealth in emerging markets result in an increase in acute and chronic diseases as well as the respective demand for effective medicines. Due to state-imposed efforts to boost generics and cut health care costs, generics now make up around 80% of the prescription drug market volume, but only account for 28% of global sales.
Number of Approved Generics and Biosimilars Increases
Due to the accelerated market approval process, the number of approved generics in the United States has grown by around 40% since 2015. A similar development is expected for biosimilars, the generic versions of biologics affected by patent expiries. In the United States, biosimilars sales and their market penetration remain modest for the time being. In an effort to reduce health care costs, however, the FDA has its mind set on changing this situation: It intends to simplify and accelerate the approval process for biosimilars. 61% of the 46 approved drugs in 2017 were subject to expedited development and review methods for accelerated market approval.
Small and medium-sized biotech companies account for a significant percentage of innovation, and they increasingly introduce their products into the market by themselves. In 2017, they made up 51% of FDA market approvals. In only 28% of FDA approvals, the large pharma companies were the originators, while inlicensing and acquisitions accounted for the remaining 21%.
New Supply Chains and Additional Services
The trend to repatriate drug substance and drug product manufacturing from Asia to the West has gained more momentum amid supply, quality, and intellectual property concerns, intensified regulatory pressure from US and European authorities, and rising costs. More than two-thirds of the drug GMP warning letters the FDA issued outside the United States over the last 5 years concerned Asian producers. The Chinese enforcement of environmental protection set to high priority by President Xi Jinping are expected to cause a reduction by half of today's chemical manufacturing plants in China. First effects of the effort are being seen along the Yangtze River causing shortages in basic chemicals and intermediates resulting in interruptions of business supply chains.
At the same time, many large pharma companies no longer regard API production as a core business area as long as it does not require proprietary technology. Small and medium-sized biotech companies are not interested in owning and tying up capital in expensive cGMP production facilities. Yet given the more stringent regulatory requirements, additional services and documentation are necessary in API development and manufacturing. This explains the growing demand for process development, process and API analytical methods, API manufacturing and documentation, as well as further-reaching services at custom development and manufacturing organizations (CDMO). CDMO also play an important role in the diversification of approval risks and quantity requirement volatility related to intensifying competitive pressure. In addition, they can support biotechs thanks to their experience in the compilation of chemical manufacturing control (CMC) documents required for the application for approval. If the required quality is delivered at the first attempt, painful opportunity costs and intense delays can be avoided in the market approval process. Reliability, an impeccable quality track record, and profound experience are the key criteria in selecting a CDMO.
CDMO Strategy Models
Currently, three basic CDMO strategy models can be observed: (i) specialization with a strong focus on a few value chain segments and niche technologies; (ii) horizontal consolidation, external growth based on capacity acquisition in the same value chain segment; and (iii) vertical integration, backward or forward integration in adjacent value chain segments, usually by acquisition. Many large CDMO with a global footprint currently execute a hybrid strategy between horizontal consolidation and vertical integration, while smaller and medium-sized CDMO tend to focus on technology and performance leadership. The real art lies in offering the entire range of development, manufacturing, and respective services for one or several value chain segments – with competent, high-quality execution and without dissipating energy. Time will tell whether (i) process and analytical development and drug substance manufacturing; (ii) drug product formulation and finishing; and (iii) the respective filling, packaging and distribution create sufficient synergistic value if they are offered, in total or partially combined, by the same company. It is clear that these three main value chain segments require completely different competences, facilities, and skills, both technologically and functionally. Offering these services from different operationally independent sites solely under the same CDMO brand does not add enough value for customers.
Quality requirements and regulations continue to increase, resulting in longer cGMP sequences in chemical API synthesis and higher cGMP manufacturing volume requirements. The increasing repatriation and outsourcing of small molecule APIs, along with the shortage of process development and manufacturing experience and capacities among biotech and pharmaceutical companies, have already created first bottlenecks in high-quality, technologically proficient chemical process development and API manufacturing capacities. This trend is set to become even more apparent over the coming few years, as many CDMO have been rattled by several changes of ownership and the subsequent restructuring or are, based on the crucial experiences over the last two decades, still unwilling to make capital-intensive investments in high-quality development and manufacturing capacities.
The topline pharma market development together with the mechanistic changes in the pharma value chain results in a promising perspective for high-quality, technologically experienced, and specialized providers of exclusive synthesis, especially in API manufacturing. In outsourcing APIs that are still under patent protection, pharmaceutical companies prefer partners that are able to provide the full range of services from chemical synthesis routefinding, chemical process and analytical development, and multi-step manufacturing to the API, including validation and stability studies. On the one hand, the partners should possess a cutting-edge development and manufacturing infrastructure, an impeccable quality track record, and a broad technology platform, while on the other hand offer profound experience in the development of chemical processes and the manufacturing of APIs. In addition, pharmaceutical companies cooperate with a select group of strategic partners throughout all stages from development up to market introduction and supply.
Contact
Dottikon Exclusive Synthesis AG
Hembrunnstr. 17
5605 Dottikon
Switzerland