Opportunities Through Biotechnology
06.06.2014 -
Latin America continues to be an area of rising investment for the pharmaceutical industry, with Brazil and Mexico leading the way. Not only is Latin America an attractive target for potential market entrants, with the help of regulatory changes and burgeoning partnerships, it solidifies its commitment to further expanding its presence on the global pharmaceutical marketplace. Keeping pace with this expansion is increasing oversight from the Brazilian regulatory authority for domestic and foreign market entrants alike.
Brazil and Latin America as a Market
With its growing population of over 200 million people and an estimated worth of $25 billion, Brazil is the largest pharmaceutical market in Latin America. Brazil surpasses second ranked Mexico by over 80 million people and nearly double the market worth. Healthcare in Brazil is a big business with 9% of gross domestic product (GDP) going to healthcare expenditures. As these costs are increasing, governments in Latin America are emphasizing the use of generic medications and pushing for local manufacturing. Pharmaceutical production is fast becoming a substantial market in Latin American countries that can provide the infrastructure needed. For example, Mexican pharmaceutical manufacturing accounts for over 15% of the country's total manufacturing GDP.
Opportunities Through Biotechnology
Biotechnology is an area that continues to see increased investment, both for active pharmaceutical ingredient (API) and finished dose manufacture. As costs for biologic and other specialty manufactured medications continue to rise, the Brazilian government has sought to expand local manufacturing capabilities. As a means to acquire technology and augment capacity affordably, there has been a trend in deal making between companies in other emerging markets.
In mid-March, local manufacturer Cristalia Laboratories received Good Manufacturing Practices (GMP) certification from the Brazilian regulatory authority, Agência Nacional de Vigilância Sanitária (ANVISA) for three biotech APIs. The approval for trastuzumab, etanercept, and somatropin marked the first domestic company to receive ANVISA GMP certification for biotech derived products. It was achieved through a two year partnership with Alteogen in South Korea. To further develop this area, Brazilian laboratory Fiocruz has entered into a partnership with Ukrainian based Indar. Indar will transfer the technology needed for Fiocruz to manufacture insulin crystals, with the expectation that Fiocruz will be operating at scale by 2016. Additionally, fellow BRIC (Brazil, Russia, India, and China) based company Biocad announced plans last year to build a biosimilar plant in Brazil with supplemental funding being supplied by Brazil's National Bank for Economic and Social Development.
Another specialty area that has seen considerable growth is that of vaccine manufacturing. Historically, vaccine manufacture was dominated by a small number of big pharma companies, but there is a growing trend of manufacturing in emerging markets. In order to enhance local supplies as well as supplement other emerging markets stocks, companies in Brazil and Latin America have been boosting production. Recently, a partnership between the Center for Genetic Engineering and Biotechnology in Cuba and newly formed French company, ABIVAX, regarding vaccine research and development was announced. If successful, the Cuban developed vaccine would be marketed in Europe and other areas.
Regulated Market Presence
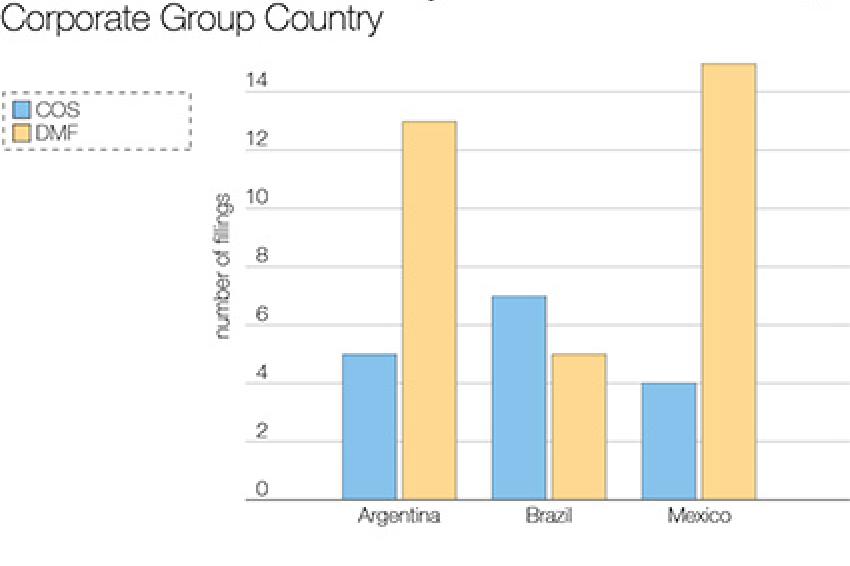
To date, corporate groups headquartered in Brazil have had a limited presence in highly regulated markets, like those of the US and EU. Utilizing data from Thomson Reuters Newport Premium, this presence can be better quantified. As of mid-May, active US drug master files (DMF) and European certificates of suitability (COS) held by corporate groups in Brazil account for 12 of the 49 active filings held by Latin American corporate groups in total (fig. 1). Additionally, following implementation of the Generic Drug User Fee Act, US DMFs that have been referenced in subsequent filings and have paid the fee are considered available for reference. No corporate groups based in Brazil appear on the FDA's list of DMFs available for reference as of mid-May. Mexico is the only Latin American country that has domestic corporate groups with DMFs available for reference. Evaluation of Brazil's API manufacturing presence compared to that of the other BRIC countries shows that, while its manufacturing base is far smaller than that of China or India, it has a higher number of manufacturers with some regulated market presence than Russia (fig. 2). Under Europe's Falsified Medicine Directive, Brazil is undergoing an equivalence assessment to join Australia, Japan, Switzerland, and the US on the list of third countries that will not need to submit written confirmations for exports to the EU.
Market Entry Challenges
Regulations among Latin American countries vary, and gaining approval from ANVISA can take significant time, two years in most cases, with some taking up to four years. Additionally, due to a substantial backlog at Brazil's patent office, obtaining pharmaceutical patents in Brazil has been known to take up to 10 years in some cases. Further adding to the patent timelines, Brazil seemingly granted ANVISA the authority to review pharmaceutical patents for patentability requirements. This review essentially allows the health agency to veto patent applications and contradicts earlier views that such discretions were not within ANVISA's scope.
While there is a greater push towards transparency, the many layers to gaining market entry in Brazil often necessitate a local partner or presence to help facilitate approval of products. Additionally, multinational companies which have purchased local companies to gain access to the domestic market often find difficulties integrating local business practices and market needs with foreign strategy.
Takeaways
The Brazilian market is growing and offers substantial prospects for companies that are able to navigate the regulatory system to bring products into this complex country. To be successful, foreign companies will need careful evaluation of market needs and to take a targeted approach for which products to bring to market. Addressing local manufacturing deficiencies and critical need products through partnerships for biotech products and manufacturing will continue to be an important growth segment for this region. Additionally, while Latin America's regulated market presence is small, it is growing and more companies are investing in facilities and partnering for capabilities, providing ample opportunities for the future.
Contact
Thomson Reuters
215 Commercial Str.
Portland, Maine 04101
+1 207 8719700
+1 207 8719800